
by Rachel Montague | Apr 14, 2022 | Industry insights
Written by Luke Parkes, Assistant Project Manager ILAP remit It has been just over a year since the Medicines and Healthcare Products Regulatory Agency (MHRA) established the Innovative Licensing and Access Pathway (ILAP) in response to Britain’s exit from the...

by Rachel Montague | Apr 4, 2022 | Industry insights
Written by Pip White, Senior Medical Writer The global value dossier (GVD) is a vital resource to ensure internal strategy alignment and facilitate external communication. As described in our previous blog, GVDs incorporate evidence-based messaging, comprehensive...

by Rachel Montague | Feb 28, 2022 | Industry insights
Written by Harry Atkins, Assistant Project Manager Today is Rare Disease Day, a day to raise awareness for people across the world living with a rare disease, their families, and those who care for them. Around 300 million people worldwide have been diagnosed...

by Rachel Montague | Dec 17, 2021 | Industry insights
Written by Amy Crompton, Systematic Review Analyst Previously, we discussed the use of real-world evidence (RWE) in the reimbursement assessment of medical devices. In this latest blog, we present an overview of the RWE framework created by the National Institute for...
by Rachel Montague | Dec 13, 2021 | Industry insights
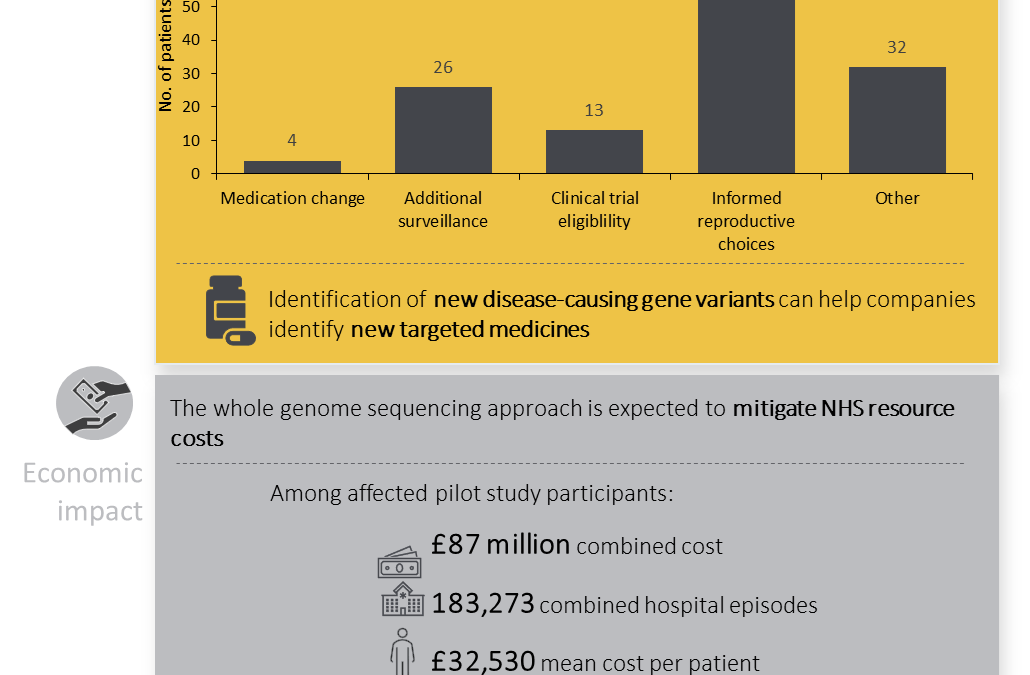
Written by Emma Lones, Medical Writer What are rare diseases? In the European Union, a rare disease is defined as a disorder affecting ≤5 in 10,000 persons (1). Using this definition, the global population prevalence of rare diseases is estimated to be between...
by Rachel Montague | Nov 30, 2021 | Industry insights
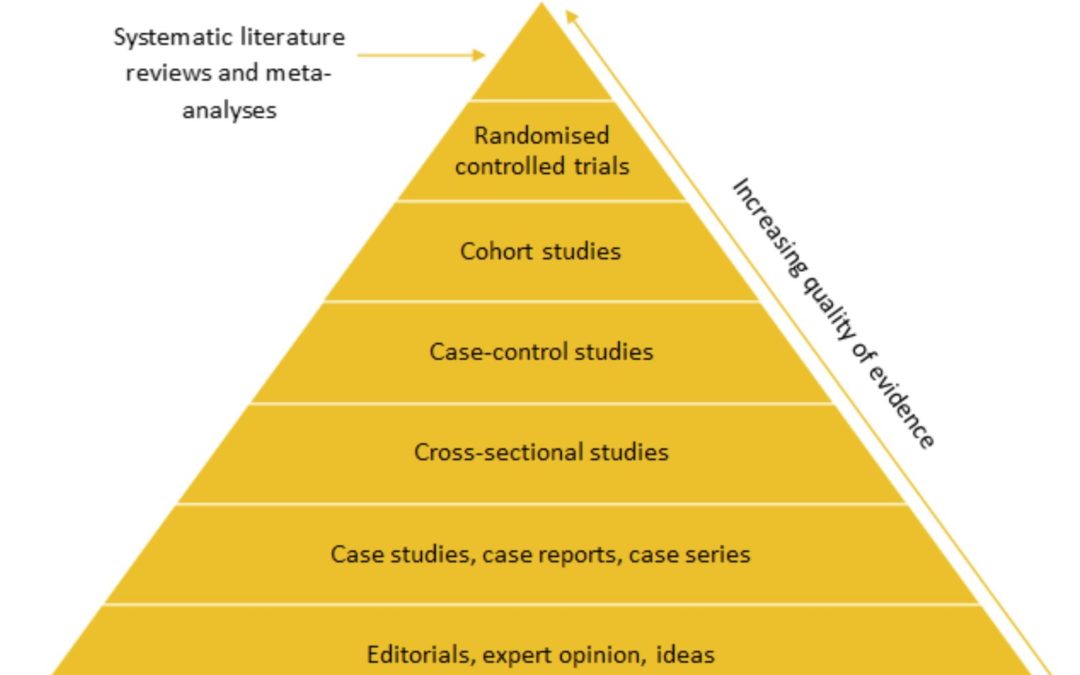
Amy Crompton (Systematic Review Analyst) and Tom Macmillan (Consultant – Systematic Review) Hierarchy of evidence Different study designs are often ranked in a hierarchy of evidence based on their validity and robustness. Randomised controlled trials...






