Written by Emma Bartram, Associate Systematic Review Analyst
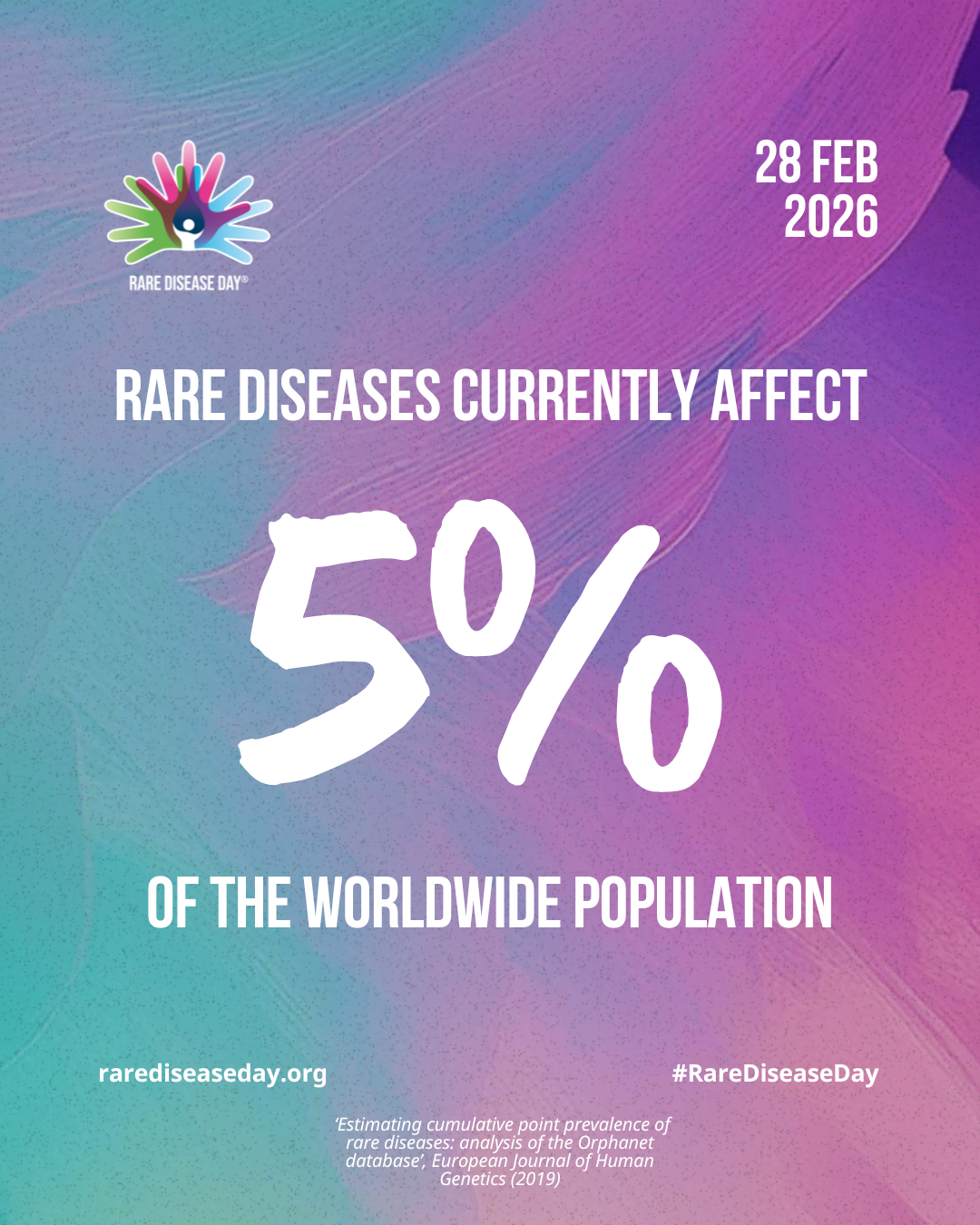
A rare disease is one that affects fewer than 1 in 2,000 individuals, and yet 3.5–5.9% of people will experience a rare disease over the course of their lifetime (1,2). Many of these rare conditions are profoundly debilitating, underscoring the urgent need for the development of novel therapies (2).
Drug development for rare diseases presents a unique set of challenges. Traditional randomised controlled trials (RCTs) often face recruitment difficulties due to small patient populations, and in therapy areas without an effective standard of care, assigning patients to placebo raises ethical concerns (3). This has led to an increasing reliance on real world evidence to support findings from clinical trials (3). Moreover, drug development is expensive, and pharmaceutical companies are not guaranteed to recoup all costs when the potential market is limited (4).
The 1983 United States (US) Orphan Drug Act (ODA) was introduced to address these challenges (3). Before its passage, approximately two rare disease drugs were approved each year in the US (5). The impact of the ODA has been significant, with almost half of US Food and Drug Administration (FDA) approvals between 2017 and 2021 being for orphan drugs (6). The ODA inspired similar initiatives worldwide, including Australia’s Orphan Drugs Program in 1997 and the European Medicines Agency’s (EMA) orphan designation scheme in 2000 (7,8). Following the United Kingdom’s (UK) departure from the European Union (EU), the Medicines and Healthcare products Regulatory Agency (MHRA) established an independent orphan designation pathway in 2021 (9).
Orphan drug designation typically requires that a condition is life‑threatening or chronically debilitating, and that the proposed therapy offers a potential significant benefit over existing treatments (7,9). Prevalence thresholds vary by authority: the UK and EMA use a cutoff of 1 in 2,000 people, whereas the US applies an absolute threshold of 200,000 individuals (6,7,9). In certain cases, exceptions may be granted for more common diseases when developers are unlikely to recover the costs of bringing a therapy to market (6,7).
Orphan drug designations are typically granted early in the drug development process, with up to 10 years of exclusivity after marketing authorisation, during which similar products for the same indication are blocked unless they demonstrate clear superiority (6,7). In the UK and EU, a further 2 years may be granted when paediatric studies are successfully completed (9, 10). Additional incentives include reduced or waived fees for regulatory procedures (such as inspections), marketing authorisation applications, scientific advice, and protocol assistance, which substantially reduce early‑stage development costs (6,7,9). In the US, the Orphan Products Grants Program also provides direct funding for early‑stage clinical research (11).
Despite significant progress in recent years, over 90% of rare diseases still lack an approved therapy (12). Treatments that do exist are often expensive, reflecting the scientific, regulatory, and commercial challenges of developing medicines for very small patient populations (4). Continued investment, regulatory flexibility, patient‑centred research, and patient advocacy remain essential to drive innovation and improve patient outcomes.
You can find out more about Rare Disease Day here.
If you are interested in finding out more about our market access services, please contact the Market Access and Value Communication team at Source Health Economics, a HEOR consultancy specialising in evidence generation, health economics, and communication.
References:
- Nguengang Wakap S et al. Eur J Hum Genet. 2020;28(2):165-173.
- Rare Diseases Day. 2025; What is a rare disease? Avaliable at:
https://www.rarediseaseday.org/what-is-a-rare-disease/ - Mellerio JE. Br J Dermatol. 2022;187(4):453-454.
- Berdud M et al. Cost Eff Resour Alloc. 2020;18:31.
- Fermaglich LJ et al. Orphanet J Rare Dis. 2023;18(1):163.
- Congressional Research Service (CRS). 2024; The Orphan Drug Act: Legal Overview and Policy Considerations. Avaliable at: https://www.congress.gov/crs-product/IF12605
- European Medicines Agency (EMA). Orphan designation: Overview. Avaliable at:
https://www.ema.europa.eu/en/human-regulatory-overview/orphan-designation-overview - Orphanet. Avaliable at: Orphan drugs in Australia. Avaliable at:
https://www.orpha.net/en/other-information/about-orphan-drugs?stapage=australia - Medicines and Healthcare products Regulatory Agency (MHRA). 2020; Orphan medicinal products. Avaliable at: https://www.gov.uk/guidance/orphan-medicinal-products-in-great-britain
- Bolislis WR et al. Clin Ther. 2019;41(12):2630-2642.
- Federal Food and Drug Administration (FDA). Orphan Products Grants Program. Avaliable at: https://www.fda.gov/industry/grant-programs-support-development-medical-products-rare-diseases/orphan-products-grants-program
- Kaufmann P et al. Orphanet J Rare Dis. 2018;13(1):196.