Written by Abby Paine, Statistician
It’s National Numeracy Day, which is run by National Numeracy, a charity that aims to enable everyone across the UK to be confident and competent in using numbers and data. This is such an important issue; you may not realise it, but we all use maths to make decisions every day; whether you are following a recipe, shopping, deciding what time to leave the house to meet a friend, or determining whether that must-have tourist souvenir in a foreign currency is overpriced or not!
I loved maths from a very young age, but I have to admit that, despite using statistics extensively in my current role, it was definitely my least favourite branch of maths at school! This all changed when I entered the world of work, and I could see how powerful statistics are in providing evidence for the benefits of new medicines.
Meta-analyses are ranked at the top of the evidence-based medicine hierarchy (Figure 1). This hierarchy can be used to rank the strength of different study designs, which is particularly important to health technology assessment (HTA) bodies, such as the UK’s National Institute for Health and Care Excellence (NICE).
Figure 1: The evidence-based medicines pyramid
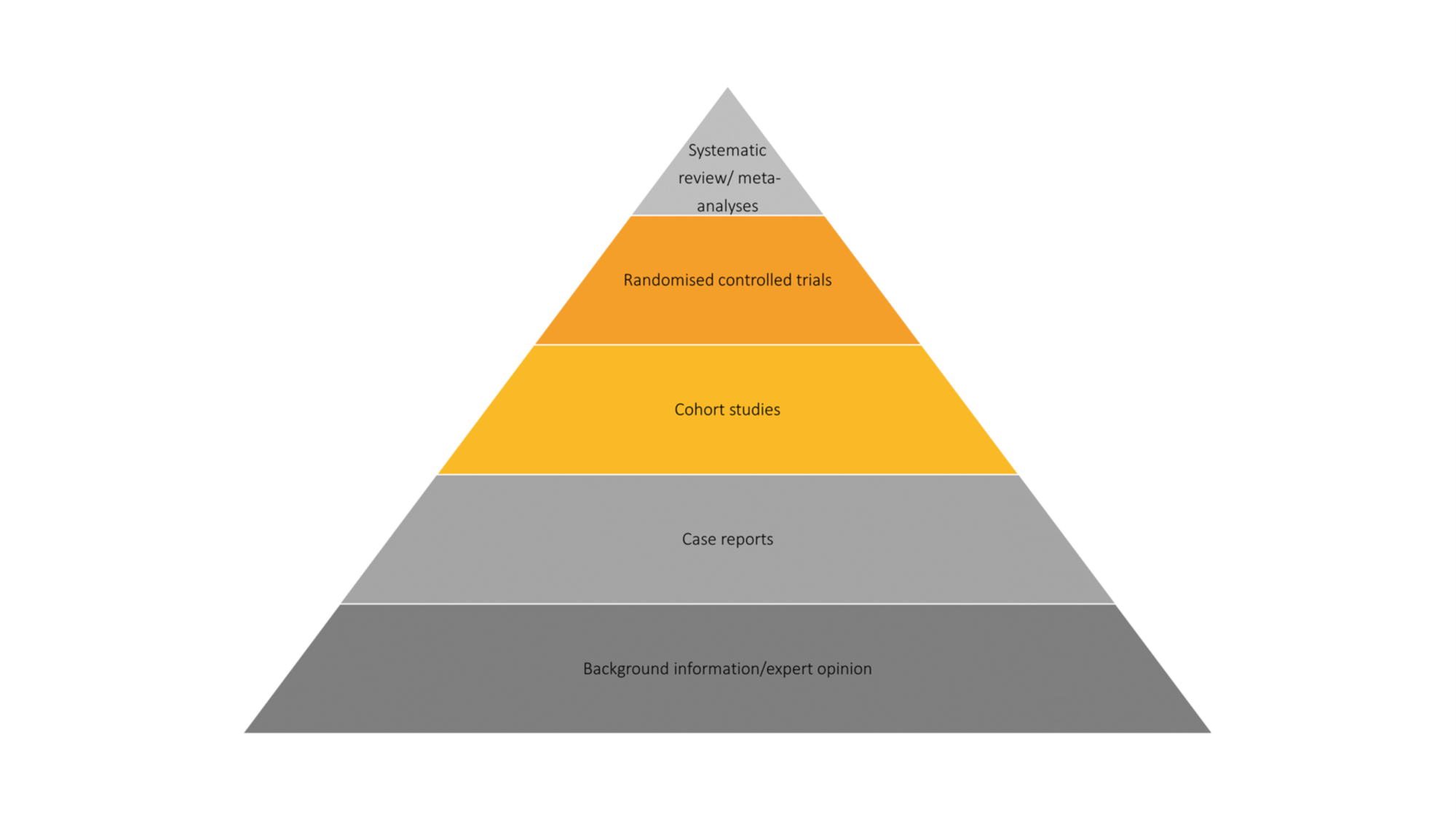
I have now spent more than 10 years working on meta-analyses, as well as two related statistical methods: indirect treatment comparisons and network meta-analyses. NICE provides the following definitions for these three methods:
- Meta-analysis: A method often used in systematic reviews to combine results from several studies of the same test, treatment, or other intervention to estimate the overall effect of the treatment
- Indirect treatment comparisons (ITCs): An analysis that compares interventions that have not been compared directly in a head-to-head, randomised trial (Figure 2)
- Network meta-analyses (NMAs): An analysis that compares two or more interventions using a combination of direct evidence and indirect evidence
Figure 2: Direct and indirect treatment comparison
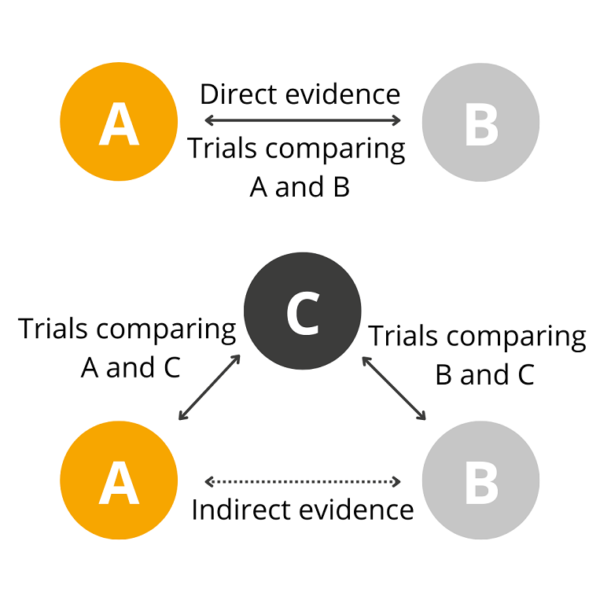
The maths behind these methods is surprisingly simple, building on what you will have learned at school during your GCSEs and A-Levels. For example, if we wanted to consider an outcome for which there are just two possible outcomes, such as response to treatment (yes/no), then we are interested in calculating an odds ratio as a measure of the relative treatment effect. For this type of data, we apply a log transformation, as this has better statistical properties to which we can apply a simple formula from A-level maths. We know that:
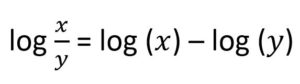
and so the indirect calculation of relative effect for treatment A vs B is:
![]()
which is the same as:
![]()
All NMAs do is build on these basic formulae to calculate many treatment comparisons from multiple trials all at the same time.
Source has conducted a number of meta-analysis, ITC, and NMA projects across a broad range of indications including oncology, endocrine disease, eye conditions, and inflammatory conditions. It is a very satisfying feeling to run the statistics and provide a favourable result for our clients’ new product in comparison with existing treatments.
Given the prohibitive cost of running a head-to-head trial with every possible comparator, NMAs and ITCs will continue to provide strong evidence of relative treatment effect for many years to come, especially to support submissions to HTA bodies.
Despite using maths every day in my job, there are still so many more things I could learn! If you are interested in developing your maths skills, why not have a look at the freely available resources on the National numeracy website and celebrate National Numeracy Day.
If you would like to learn more about the statistical or evidence synthesis capabilities within our team, or about the bespoke training we offer, please contact us at Source Health Economics, an independent consultancy specialising in evidence generation, health economics, and communication.




