Thank you for visiting us at booth 815

Evidence Generation Contact Neil Webb
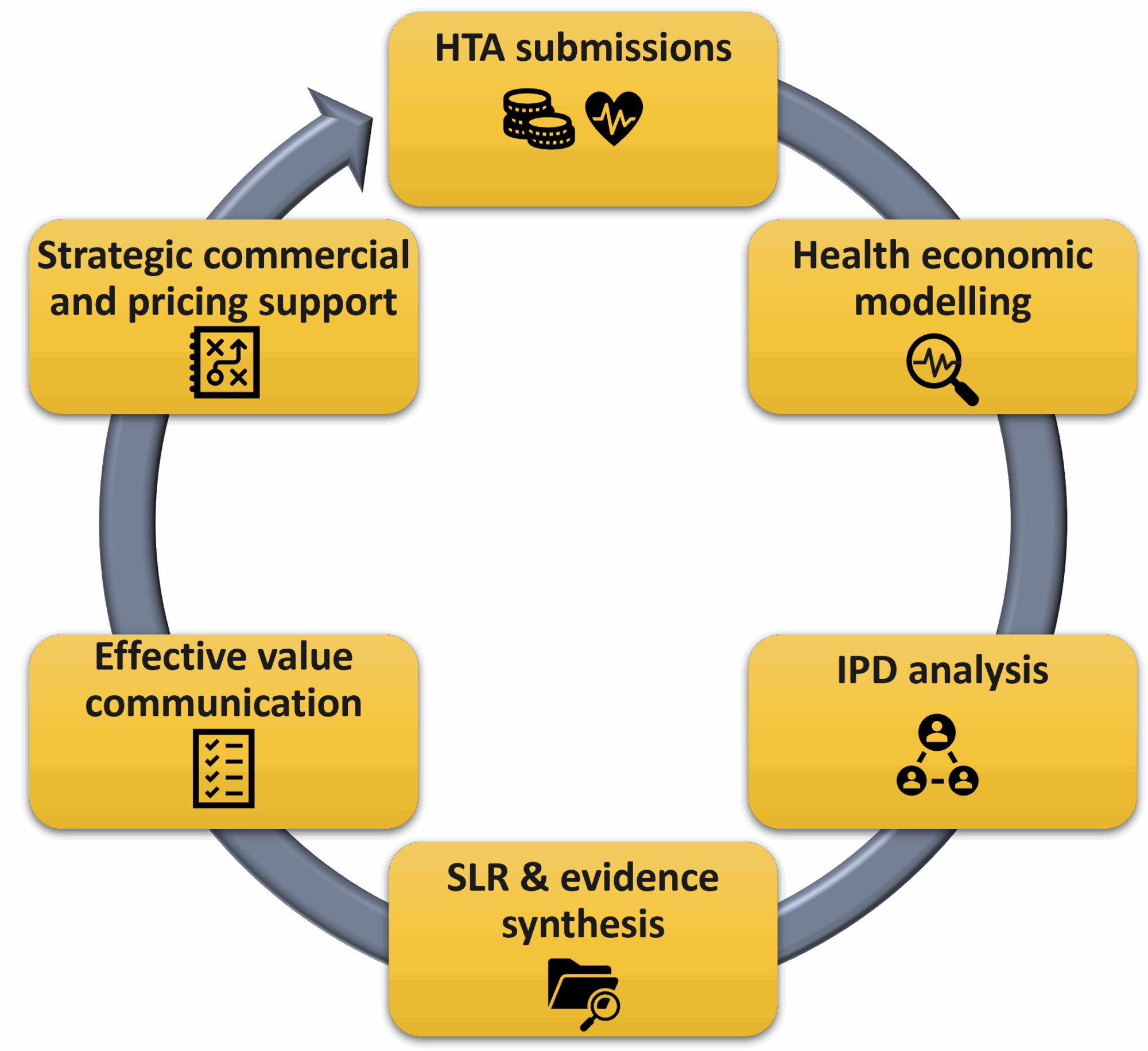
Our systematic review team produces high-quality, user-friendly deliverables:
- Systematic literature reviews
- Targeted literature reviews
- Indirect treatment comparisons
Click here to view our team credentials
Economic Modelling Contact: Alec Miners
We provide design, development, and strategic review of economic models
- Cost-effectiveness models
- Budge-impact models
- Individual patient data analysis
- Advisory boards
- Training
Click here to view our team credentials
HTA and Client Services Contact: Peter O’Neill
We support HTA submission development to UK and international agencies, with strategic advice throughout the process
- HTA submissions to European and North American reimbursement agencies
- HTA strategy
- HTA advisory boards
- Mock committee meetings
Click here to view our team credentials
Market Access and Value Communications Contact: Deb Burford
Our medical writers develop clear and concise written materials reflecting an evidence-based value proposition
- Evidence hierarchies
- Global value/reimbursement dossiers
- Value message development
- Slide decks, brochures, flashcards
- Payer communication tools
- Publications
- App content development
Click here to view our team credentials
Commercialisation and Pricing Contact: Jo Wallis
We support companies with the development and implementation of commercialisation strategies to optimise value for our clients from Phase I to reimbursement success:
- Integrated pricing and early commercial access strategy development
- Commercial negotiation support
- Pricing analysis and research, including payer landscape assessments
Click here to view our team credentials
1. Comparing Risk of Bias Assessment Requirements Between the EU JCA and European HTA Agencies. 10th November, Poster Session 2, Poster number HTA83.
2. Similarities and Differences Between the NICE and EU JCA Clinical SLR Requirements.11th November, Poster Session 4, Poster number HTA298.

SCIRIS brings together 5 outstanding agencies with a team of over 300 people who are driven to deliver exceptional results in communications and consulting. Operating globally, with offices in North America and the United Kingdom, we offer best in class capabilities across multiple therapeutic categories and all phases of commercial and brand development, offering bespoke programmes in healthcare communications, creative and brand strategy, medical compliance, HEOR consultancy, and insights
Porterhouse Medical Group
Porterhouse Medical is a global, strategic, scientific communications group providing powerful, insight-driven, omnichannel communication services to the pharmaceutical industry worldwide. Click here to find out more
Porterhouse Insights
Porterhouse Insights is a global research consultancy driven to generate actionable insights to meet the evolving needs of our biopharmaceutical clients. The attached infographic is an introduction to Porterhouse Insights, SCIRIS’ global strategic research, insights and evidence consultancy, and the touchpoints across the R&D and commercialisation process where the agency offers support to their clients.
Indigo Medical
Indigo Medical Communications combines strategy, science, skilled writing, and creativity to deliver solutions that anticipate client needs. Click here to find out more
Would you like more information?

Juliet Warner
Director

David Trueman
Director











